In a separate announcement made today, the Company also secured A$83 million in private placement funding to support growth initiatives, including European expansion.
MELBOURNE, Australia, March 31, 2026 /PRNewswire/ — 4DMedical Limited (ASX:4DX), the global leader in cardiothoracic imaging software, announces that its latest imaging technology, CT:VQ™ has received CE Mark certification for commercial use in the European Union. 4DMedical will quickly launch commercial deployment of CT:VQ across one of the world’s largest respiratory imaging markets.
In a separate but concurrent announcement, 4DMedical has secured firm commitments from select institutional investors for an AU$83 million single-tranche private placement. This additional capital will support the Company’s growth strategy, particularly the expansion of CT:VQ™ in Europe.
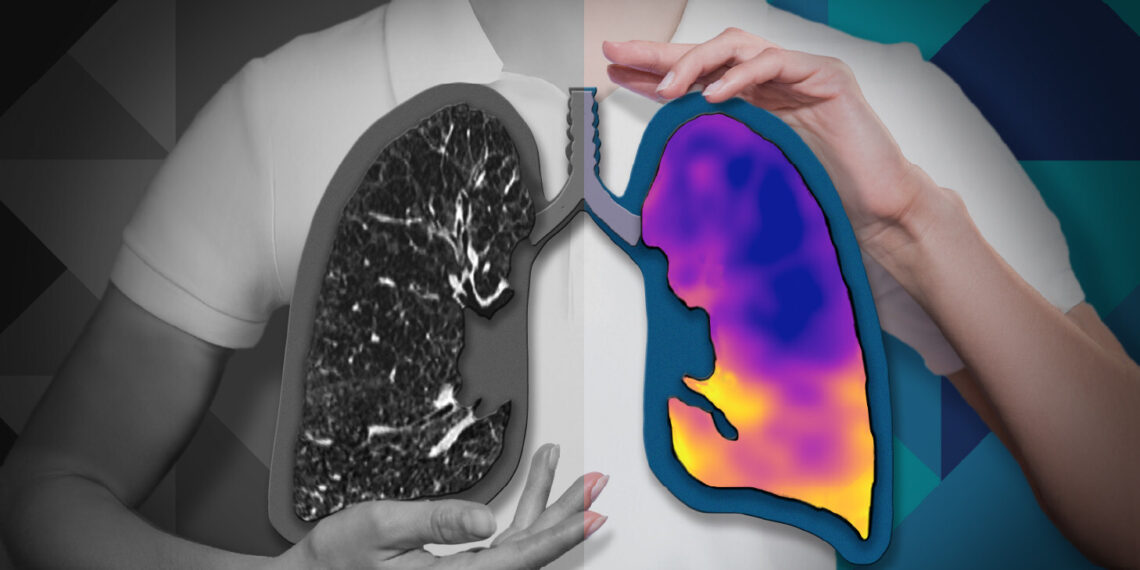
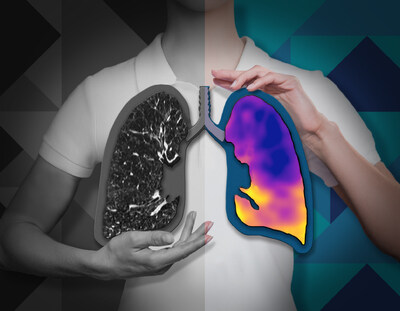
CT:VQ™ is the world’s first and only non-contrast, ventilation-perfusion imaging technology, providing quantitative functional lung insights from routine non-contrast CT scans without the need for radiotracers or specialised nuclear medicine infrastructure.
Pulmonary disease is fundamentally functional—defined by abnormalities in ventilation and perfusion—yet most routine imaging available to radiologists and pulmonologists remains purely structural. CT:VQ™ bridges that gap by generating ventilation and perfusion maps allowing clinicians to visualize regional lung function with the anatomical clarity of CT.
For large medical institutions, CT:VQ™ is designed to fit within existing CT-based workflows and infrastructure. CT:VQ offers relief from long-standing constraints associated with conventional nuclear VQ imaging, including radiotracer availability, workforce limitations and operational complexity.
The European market represents a significant opportunity for advanced cardiothoracic imaging. With a population of more than 450 million, a highly developed hospital-based imaging infrastructure and an extensive installed base of CT scanners, the EU is well positioned for adoption of CT-based functional lung imaging. 4DMedical estimates that approximately 400,000 nuclear VQ scans are performed annually across the EU, underscoring the scale of clinical need.
“CE Mark certification for CT:VQ™ is a significant milestone that opens access to one of the world’s largest and most sophisticated healthcare markets,” said Andreas Fouras, Managing Director, CEO and Founder of 4DMedical. “Combined with FDA clearance, 4DMedical now has regulatory clearance to rapidly commercialise CT:VQ™ across both the U.S. and the EU.
“The clinical need for CT:VQ™ is universal. The limitations of nuclear VQ scanning—radiotracer constraints, limited access and operational complexity—exist across healthcare systems globally. Europe has the clinical expertise, imaging infrastructure and research leadership to play a major role in the adoption and evidence generation for this technology.”
The CE Mark follows growing U.S. momentum for CT:VQ™, which is now deployed at six leading academic medical centres: Stanford, Cleveland Clinic, University of Miami, UC San Diego Health, University of Chicago Medicine, and the recently announced initial deployment at Mayo Clinic. 4DMedical believes this early adoption by major U.S. institutions demonstrates strong interest in CT:VQ™ among leading radiology and pulmonary teams and provides a blueprint for expansion into Europe.
4DMedical said it is now advancing plans to expand commercial engagement across Europe and to work with leading hospitals and clinicians on adoption, evaluation and research initiatives for CT:VQ™.
About CT:VQ™
CT:VQ™ is 4DMedical’s proprietary ventilation-perfusion imaging solution that generates functional lung information from non-contrast CT scans. Built on the Company’s patented XV Technology®, CT:VQ™ is designed to provide quantitative ventilation and perfusion insights within familiar CT workflows, without requiring radiotracers.
About 4DMedical
4DMedical Limited (ASX:4DX) is a global medical technology company that creates and deploys the most advanced software-based cardiothoracic imaging technology—integrating both proprietary algorithms and artificial intelligence. 4DMedical’s software platform delivers deep, quantitative, insights from routine clinical imaging to help clinicians assess lung function and cardiopulmonary disease.
These insights help respiratory physicians, radiologists and hospital teams quantify regional lung function, support diagnosis and disease monitoring, and inform treatment and surgical planning. 4DMedical’s solutions are designed to integrate into existing clinical workflows. They provide actionable, patient-specific information that supports better decision-making and operational efficiency across a range of respiratory conditions. Learn more at www.4dmedical.com.

Photo – https://mma.prnewswire.com/media/2944827/Lungs_Graphic_Cutouts_Color_BW.jpg
Logo – https://mma.prnewswire.com/media/2730645/5888947/4DMedical_Logo.jpg
![]() View original content:https://www.prnewswire.com/apac/news-releases/4dmedical-receives-ce-mark-for-ctvq-enabling-commercial-launch-across-the-european-union-302727841.html
View original content:https://www.prnewswire.com/apac/news-releases/4dmedical-receives-ce-mark-for-ctvq-enabling-commercial-launch-across-the-european-union-302727841.html
SOURCE 4DMedical