SHANGHAI, Jan. 8, 2026 /PRNewswire/ — Shanghai Ark Biopharmaceutical Co., Ltd. (“ArkBio”) today announced that China’s National Medical Products Administration (NMPA) has approved the New Drug Application for Serdexmethylphenidate Chloride and Dexmethylphenidate Hydrochloride Capsules (Aizhida) for treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years of age and older in China.
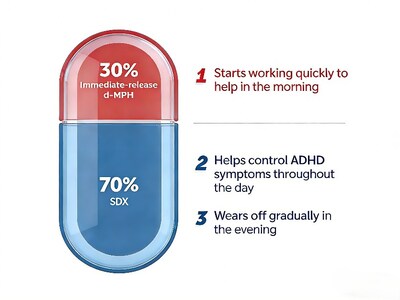
Aizhida is a central nervous system stimulant indicated for the treatment of ADHD. Each once-daily capsule contains serdexmethylphenidate (SDX), a prodrug of dexmethylphenidate, co-formulated with immediate-release dexmethylphenidate (d-MPH) in a fixed ratio designed to provide both rapid and extended symptom control.After ingestion, the immediate-release d-MPH component is rapidly absorbed to provide early onset of effect, while SDX is gradually converted to d-MPH in the lower gastrointestinal tract, supporting sustained therapeutic exposure throughout the day. Aizhida was approved by the U.S. FDA in March 2021 as a once-daily treatment option for patients with ADHD 6 years of age and older.
ADHD is a common, chronic neurodevelopmental disorder that typically begins in childhood and may persist into adulthood. It is primarily characterized by age-inappropriate levels of inattention and/or hyperactivity-impulsivity. In China, the prevalence of ADHD in children and adolescents is estimated at approximately 6.4%, affecting over 23 million individuals. Studies have shown that symptoms persist into adolescence for 60%-80% of patients, and into adulthood for roughly half of cases. Current ADHD treatment options in China are limited, and currently available medications do not fully meet clinical needs in terms of both efficacy and safety for many patients. Some patients discontinue treatment due to suboptimal response, side effects, or dosing inconvenience, highlighting the need for additional therapeutic options.
Robust Clinical Data Addresses Unmet Need: In a pivotal Phase III clinical trial in Chinese patients with ADHD, Aizhida met both the primary and key secondary endpoints. The study demonstrated statistically significant and clinically meaningful improvements in core ADHD symptoms compared to placebo at all assessment time points. With limited treatment options in China and historical supply constraints for traditional single-agent therapies, the approval of this innovative combination product offers an important new choice for clinicians and patients.
Unique Dual Mechanism for Rapid Onset and All-Day Coverage: Aizhida combines immediate-release and prodrug-based extended-release technologies. Its ‘rapid onset + full-day coverage’ profile positions it to reshape the ADHD treatment paradigm in China. As the first ADHD medication in China to deliver both rapid onset and sustained symptom control, it fills a key clinical gap and has the potential to become a preferred treatment option.
Professor Yi Zheng, Chief Expert at Beijing Anding Hospital, Capital Medical University, and Lead Principal Investigator of the Aizhida Phase III trial in China, commented: “ADHD is one of the most common neurodevelopmental disorders in childhood, with potential impact lasting into adulthood, posing long-term challenges to learning, social functioning, and family life. While the prevalence in Chinese children is around 6.4%, treatment options—especially innovative ones balancing efficacy and safety—remain insufficient. The approval of Aizhida provides clinicians with a new therapeutic tool. We anticipate its real-world application will help improve the treatment landscape for ADHD patients in China, offering new hope particularly for those with suboptimal response or tolerance to existing therapies.”
Professor Jing Liu, Director of the Child Psychiatry Center, Peking University Sixth Hospital, and Lead Principal Investigator of the Aizhida Phase III trial in China, added: “We are committed to advancing standardized diagnosis and treatment for pediatric mental disorders in China. Comprehensive intervention is essential for ADHD, with pharmacotherapy being a foundational component. The approval of a new drug not only increases options but also prompts deeper reflection on optimizing treatment strategies. We hope the approval of Aizhida will enrich ADHD treatment approaches and contribute to better long-term outcomes, which is the ultimate goal of our clinical research.”
About ArkBio
ArkBio is a global biotech company focused on developing innovative therapeutics for respiratory and pediatric diseases. Founded in 2014, it has built core technology platforms and a differentiated R&D pipeline through in-house R&D efforts and external collaboration. Key drug assets include ziresovir (AK0529), the first direct-acting RSV antiviral with positive pivotal phase III results, and AK3280, a potentially best-in-class drug with positive phase 2 results to treatment idiopathic pulmonary fibrosis. Another core product Aizhida (AK0901) was recently approved in China for ADHD and has now entered the commercial launch phase.
ArkBio has established strategic partnerships with several multinational pharmaceutical companies and academic institutes, including Roche, Genentech, the Scripps Research Institute, the Institute of Microbiology of Chinese Academy of Sciences, domestic and international biotechnology companies, as well as venture capital institutions.
For more information, please visit: www.arkbiosciences.com
Investor Inquiries: [email protected]
![]() View original content to download multimedia:https://www.prnewswire.com/apac/news-releases/ark-biopharmaceutical-receives-china-marketing-authorization-for-aizhida-for-the-treatment-of-adhd-302656314.html
View original content to download multimedia:https://www.prnewswire.com/apac/news-releases/ark-biopharmaceutical-receives-china-marketing-authorization-for-aizhida-for-the-treatment-of-adhd-302656314.html
SOURCE Arkbio