Australia’s Therapeutic Goods Administration (TGA), responsible for approval of the safety, quality, and efficacy of therapeutic goods, has determined that Skyhawk’s SKY-0515 orally-administered small molecule therapy for the treatment of Huntington’s disease meets TGA eligibility criteria for registration via the provisional approval pathway.
Skyhawk delivered its application for provisional approval to the TGA today.
More than 115 patients are now on trial with SKY-0515.
BOSTON, March 3, 2026 /PRNewswire/ — Skyhawk Therapeutics, Inc., a clinical-stage biotechnology company developing novel small molecule therapies designed to modulate critical RNA targets for a series of challenging neurological diseases, announces that Australia’s Therapeutic Goods Administration (TGA), responsible for the approval of the safety, quality, and efficacy of therapeutic goods, has determined that the Company’s SKY-0515 orally-administered small molecule therapy for the treatment of Huntington’s disease meets TGA eligibility criteria for registration via the provisional approval pathway.

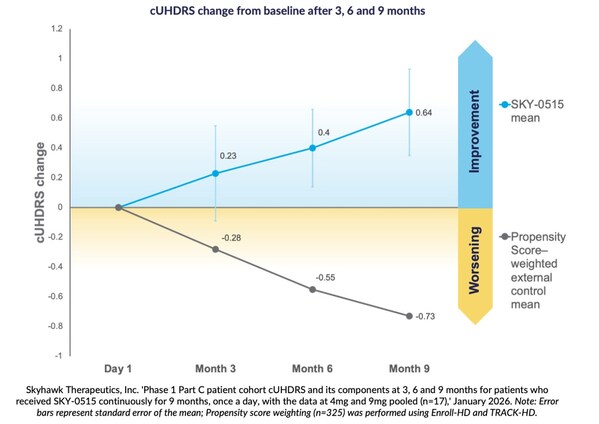
Skyhawk Therapeutics, Inc. ‘Phase 1 Part C patient cohort cUHDRS and its components at 3, 6 and 9 months for patients who received SKY-0515 continuously for 9 months, once a day, with the data at 4mg and 9mg pooled (n=17),’ January 2026. Note: Error bars represent standard error of the mean; Propensity score weighting (n=325) was performed using Enroll-HD and TRACK-HD.
Skyhawk delivered its application submission to the TGA for provisional approval today.
“We are grateful that the TGA has determined SKY-0515 for the treatment of Huntington’s disease meets its eligibility criteria for potential provisional approval,” said Bill Haney, Skyhawk’s CEO. “This represents an important first step toward what could be an accelerated path to approval in Australia and the world, offering the possibility of a disease modifying treatment for Huntington’s patients who urgently need therapies for this devastating disease.”
Treatment with SKY-0515 results in dose-dependent reductions of mHTT and PMS1, key drivers of Huntington’s disease pathology. SKY-0515 has demonstrated excellent central nervous system exposure and been generally safe and well tolerated.
Patients receiving SKY-0515 in the Phase 1C trial of SKY-0515 demonstrate mean Composite Unified Huntington’s Disease Rating Scale (cUHDRS) improvement from baseline. At nine months this improvement is +0.64 points in a pooled analysis, compared to expected worsening at nine months of cUHDRS in symptomatic patients of -0.73 points, based on propensity score weighting using Enroll-HD and TRACK-HD.
Huntington’s disease is a rare, inherited and ultimately fatal neurodegenerative disorder that affects hundreds of thousands of people worldwide, including more than 40,000 symptomatic patients in the United States and over 2,000 Australians.1,2 There are currently no approved disease-modifying treatments which slow or halt disease progression.2
SKY-0515 is a small molecule RNA splicing modifier developed through Skyhawk’s novel RNA-splicing platform, SKYSTAR®.
SKY-0515 is the first Skyhawk drug in clinical trials.
Skyhawk is developing additional novel drugs to treat neurological diseases for which there are no approved disease-modifying therapies.
About SKY-0515’s Phase 1 Clinical Study
SKY-0515’s Phase 1 clinical trial is a first-in-human trial designed to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of SKY-0515 in healthy volunteers and individuals with Huntington’s disease (HD). The trial is separated into three parts. Parts A and B evaluated SKY-0515 in Healthy Volunteers. Part C is a double-blind placebo-controlled parallel design study of two dose levels of SKY-0515 and placebo in individuals with HD (HD-ISS Stage 1, 2, or mild Stage 3) for 84 days followed by a 12 month extension of active treatment where all participants receive either a low or high dose of SKY-0515 in a blinded fashion. The objectives of the study include evaluating mutant HTT protein and PMS1 mRNA. The first patients were dosed in SKY-0515’s Part C in January 2025. Enrollment in Phase 1C of the SKY-0515 trial is now complete and the trial is ongoing.
About SKY-0515’s Phase 2/3 FALCON-HD Clinical Study
FALCON-HD (NCT06873334) is a Phase 2/3 randomized, double-blind, placebo-controlled, dose ranging study to evaluate the pharmacodynamics, safety, and efficacy of SKY-0515 in 120 participants with Stage 2 and early Stage 3 HD across 12 sites in Australia and New Zealand, and 400 participants with Stage 2 and early Stage 3 HD in 40+ worldwide sites. Eligible patients will receive a once-daily oral dose of SKY-0515 at one of three dose levels or placebo, for a treatment period of at least 12 months. This trial aims to confirm the potential of SKY-0515 to modulate RNA splicing and reduce mHTT and PMS1 proteins, which are implicated in the pathology of Huntington’s disease, and to assess the efficacy of the SKY-0515 in affecting the rate of Huntington’s disease progression. Additional information about FALCON-HD, including participating sites and eligibility criteria, can be found at ClinicalTrials.gov and www.FALCON-HD.com.
About Skyhawk Therapeutics
Skyhawk Therapeutics is a clinical-stage biotechnology company which uses its proprietary platform, SKYSTAR®, to discover and develop small molecule RNA modulating therapies for the world’s most intractable diseases. For more information visit www.skyhawktx.com.
Skyhawk Contact
Maura McCarthy
Head of Corporate Development
[email protected]
References
- Australian Institute of Health and Welfare (AIHW) 2025, Huntington’s disease in Australia, viewed 8 February 2026, https://www.aihw.gov.au/reports/neurological-conditions/huntingtons-disease-in-australia
- Huntington’s Disease Society of America (HDSA) 2026, Overview of Huntington’s Disease, viewed 8 February 2026, https://hdsa.org/what-is-hd/overview-of-huntingtons-disease/