|
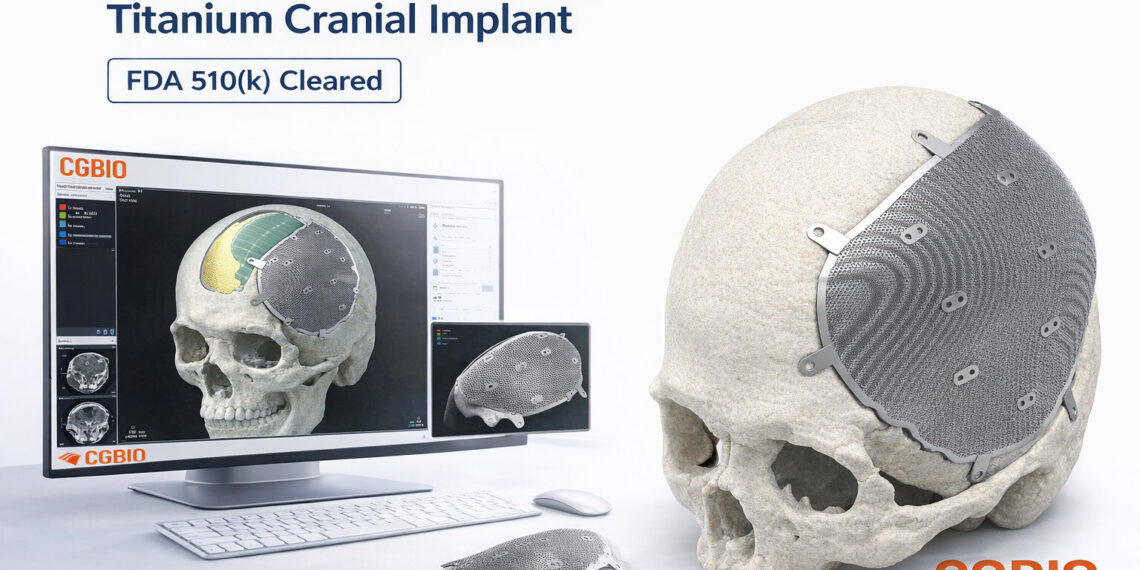
- Patient-specific titanium implant designed for cranial and craniofacial reconstruction
- First Korean company to obtain FDA 510(k) clearance for a customized titanium implant in the U.S. market
SEOUL, South Korea, April 24, 2026 /PRNewswire/ — CGBIO (CEO: Hyunseung Yu) announced that its patient-specific titanium implant, EASYMADE TI, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) pathway is a premarket clearance process that demonstrates substantial equivalence to a legally marketed predicate device, allowing commercialization in the United States.
This milestone marks the first case of a Korean company obtaining FDA 510(k) clearance for a patient-specific titanium implant intended for the U.S. market. It highlights CGBIO’s technological capabilities as well as its regulatory expertise in global markets. The achievement is particularly notable in the field of patient-specific cranial and craniofacial reconstruction.
EASYMADE TI is a patient-specific titanium implant designed based on individual CT data and intended for reconstruction of cranial and non-load-bearing craniofacial defects. The implant is manufactured using medical-grade titanium alloy and produced through Laser Powder Bed Fusion (LPBF), an advanced metal 3D printing technology.
The product delivery process is also a key differentiator. Once patient CT images are provided by U.S.-based clinicians, customized design is completed at CGBIO’s design center in Korea, and the finished implant is delivered to hospitals in the U.S. within five days. After sterilization at the hospital, the implant can be used directly in surgical procedures.
The company expects this clearance to serve as a turning point for expanding its patient-specific implant business in the United States. Demand for cranial and craniofacial reconstruction continues to grow in the U.S., driven by cases such as trauma, tumor resection, and decompressive craniectomy. At the same time, interest in patient-specific reconstruction solutions tailored to individual anatomy is increasing.
Hyunseung Yu, CEO of CGBIO, said, “The FDA 510(k) clearance of EASYMADE TI is a meaningful achievement that demonstrates CGBIO’s global competitiveness in patient-specific reconstruction solutions. We will use this milestone as a foundation to expand our customized cranial and craniofacial implant business in the U.S. and accelerate regulatory approvals and commercialization of our pipeline products.”
CGBIO plans to actively expand its presence in the U.S. market based on this clearance, while continuing to strengthen its global position in the field of patient-specific medical devices.